Date: 2/10/2024
Read Time: 5 minutes
Author: Dr Surani McCaw, B.E. (Chemical), Ph. D.

Key points:
- While effective in preventing scalding, thermostatic mixing valves (TMVs) can create conditions conducive to Legionella growth.
- Maintaining water temperatures below 20°C or above 60°C is crucial for Legionella control.
- TMV installation should be guided by a risk assessment of scalding versus Legionella infection.
- TMVs in endoscopy settings often face challenges due to installation location, flow rates, and auxiliary equipment limitations.
- Using softened water with TMVs and cartridge filters can increase microbiological risks, due to reduced chlorine content and bacteria proliferation within the softener resin.
Introduction
Thermostatic mixing valves (TMVs) are designed to mitigate the risk of scalding from hot water in healthcare settings, with the recommended safe temperature range being between 38°C and 46°C. However, this temperature range, while effective in preventing scalding, also creates an environment conducive to the proliferation of Legionella bacteria in water systems.
Several sources document the temperature profile of Legionella in water systems. Two such references are:
- UK – The Health & Safety Executive Approved Code of Practice and Guidance on Regulations (HSE ACOP L8 2013).
- Australia – The Environmental Health Standing Committee (enHealth) Guidelines for Legionella Control in the Operation and Maintenance of Water Distribution Systems in Health and Aged Care Facilities.
According to the 4th edition of HSE ACOP L8 2013:
- Legionella bacteria die at temperatures above 60°C.
- They proliferate between 20°C and 45°C.
- At temperatures below 20°C, Legionella bacteria do not multiply but can still pose a potential threat within the system.
Based on these facts, it is crucial to maintain water temperatures either below 20°C or above 60°C with effective control measures to mitigate the risk of Legionella proliferation. While maintaining hot water systems at temperatures above 60°C effectively reduces Legionella bacteria count, it also increases the risk of scalding.
This highlights the challenge of effectively managing both scalding risks and Legionella control. However, when used in accordance with applicable legislation and guidance documents, TMVs can be an effective solution. They help manage these risks by ensuring that water temperatures are kept within safe limits, thereby protecting occupants in healthcare facilities from scalding while also minimising the risk of Legionella proliferation.
The importance of risk assessment
Key notes from the ACOP L8 2013 (4th edition) guidance document emphasise the importance of risk assessment as a fundamental approach in managing Legionella.
It states that the installation and use of TMVs should be guided by a comparative risk assessment of scalding versus Legionella infection. If the assessment indicates that the scalding risk is minimal, TMVs may not be necessary.
TMV Installation
Some relevant points for TMV installation include:
- Installation Location: TMVs should be integrated directly into the tap fitting, with mixing occurring at the point of outlet.
- Proximity: TMV valves should be installed as close to the point of use (POU) as possible to minimise the storage of blended water.
- Flow Rate: Caution is needed with TMVs fitted with low flow rate spray taps on hand wash basins, as this can increase the risk.
Endoscopy Applications
The following risks are apparent in endoscopy applications:
Installation Location and Proximity
TMVs in endoscopy settings are frequently installed more than 100 meters from the point of use. It’s crucial to note that the point of use refers to the inlet point of the Automated Endoscope Reprocessor (AER), not necessarily the AER itself. In an AER, the pipework leading to this inlet point can exceed 100 meters. Majority of these installations typically operates on a demand basis rather than a recirculation system and is infrequently flushed, resulting in stagnant water during periods when the AER is not in use. This stagnant water is what the guidance documents refer to as “blended water.” According to guidance documents, TMV valve should be installed as close to the POU as possible to minimise the storage of blended water and reduce the potential for Legionella growth.
Flow Rate
In essence, the guidance documents emphasise that TMVs should only be used in applications where a risk assessment identifies a significant risk of scalding. Despite this guidance, many healthcare facilities do not fully comply with these recommendations.
Fact: A POU 0.1micron filter installed within each AER may not effectively mitigate the risk of Legionella. The risk when exchanging a filter loaded with Legionella is just as high as the risk from water coming out of a non performing filter.
In essence, the guidance documents emphasise that TMVs should only be used in applications where a risk assessment identifies a significant risk of scalding. Despite this guidance, many healthcare facilities do not fully comply with these recommendations.
Common Failures
Some common failures in practice include:
- TMVs installed before cartridge filters to maintain water temperatures between 35°C and 45°C for feeding AERs. In this setup, TMVs are integrated into the process of maintaining temperature for AERs, which are used for reprocessing endoscopes, rather than to mitigate scalding risks. This configuration often includes deadlegs or stagnant water, which can serve as a high nutrient environment for environmental pathogens such as Legionella and Pseudomonas aeruginosa.
- Water treatment companies often claim that cartridge filters with filtration ratings of less than 0.2 microns can safeguard against environmental pathogens. However, these filters are primarily designed to reduce sediments rather than microorganisms. The design of the water flow path through these cartridges itself can create conditions that exacerbate the problem by increasing deadlegs and stagnant areas, thus promoting the growth of harmful pathogens.
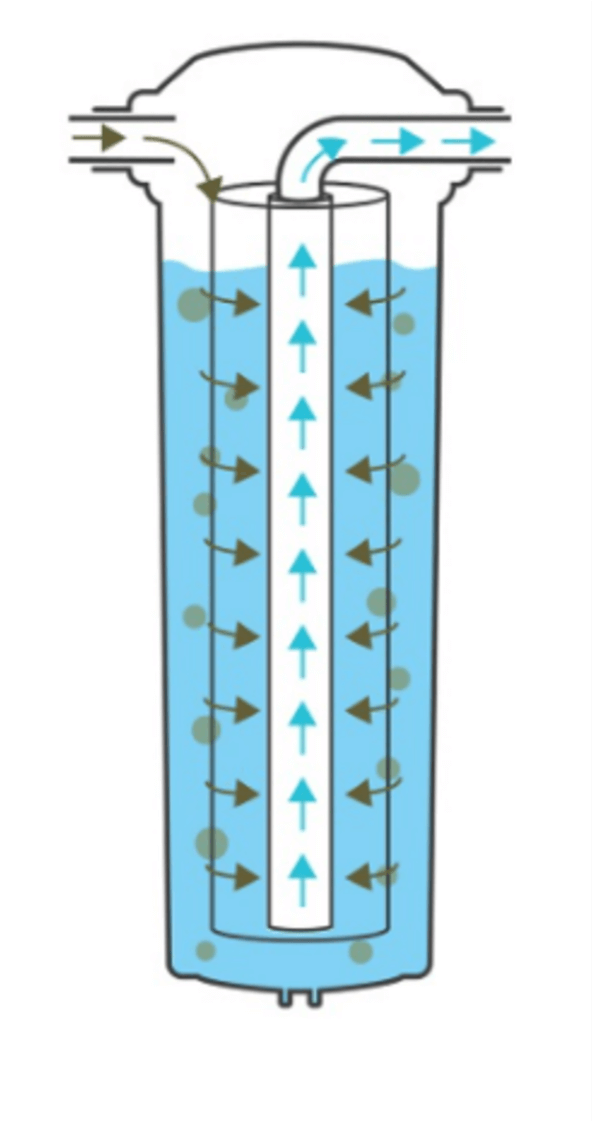
How cartridge filters operate
- Feed water enters the cartridge filter housing via an inlet port located at the same elevation as the outlet port.
- Filtered water travels through the centre tube of the cartridge filter and exits via the outlet port. With this design, as water takes the path of least resistance, water is filtered from the bottom up, which leads to the formation of stagnant water zones over time.
- These stagnant zones occur both with unfiltered and filtered water, increasing the risk of bacterial growth from both sides of the filter.
- The filter surface remains immersed in unfiltered water. This allows bacteria to migrate from the dirty side to the clean side via biofilm colonies, potentially contaminating the filtered water that exits the cartridge filter.
Microbial Risks
In addition to these risks, the use of softened water to feed TMVs presents additional microbiological risks that must be considered.
Heightened microbiological risks are:
1. Bacterial Proliferation in Softening Resins:
Water softening resins offer an ideal environment for bacterial growth. They provide a nutrient rich substrate, ample surface area, and often maintain temperatures that favour the proliferation of environmental pathogens. As a result, increased levels of bacteria, including Legionella and Pseudomonas aeruginosa, can be found in the water after passing through the water softeners.
2. Low Chlorine Content:
To extend the lifespan of the softener resin, the feed water is typically maintained at low chlorine levels. This reduced chlorine content can promote bacterial growth within the softener itself. Despite the Australian Drinking Water Guidelines (ADWG) recommending chlorine levels between 0.2 mg/L and 0.5 mg/L, the combination of increased chlorine demand from the conditions mentioned above and elevated temperatures (35°C to 45°C) can result in reduced chlorine levels after the water softener. Consequently, this may not provide adequate protection against biofilm growth in the downstream equipment and pipework.
3. Conditions in Hot Water Units:
When softened cold water is heated in hot water units, it can create conditions that are highly favourable to bacterial growth. Hot water systems that incorporate storage tanks and recirculation loops may not always adhere to optimal hydraulic conditions necessary to minimise bacterial proliferation. Issues such as improper temperature differentials, insufficient flow velocities, and excessive deadlegs contribute to the formation of biofilm and contamination of TMVs.
In Conclusion
Continuing to use town water with TMVs and cartridge filters designed for sediment reduction in endoscopy applications is not advisable given the microbiological risks. It’s essential to evaluate and potentially revise your water treatment and filtration strategy to ensure effective management of these risks, protecting both staff and patients.
Have questions?
Contact our team for answers. We’re here to address your questions and provide the solutions you need. Call 1800 656 771 or email Surani.